
Let's do another example, and again, I'll start with the wrong way to do it, and then we'll talk about the correct way. Resonance structures, because they get in the way, and once you understand what's going on it's not necessary toĭraw in those hydrogens. 
That's why we leave off those hydrogens when we\re drawing our Let me go ahead and draw it again on the right just for clarity. Hydrogens drawn in there, which is why we leave them off. Now, it looks a little bit confusing when I have those So one, two, three and four, so now the formal charge is zero. Plus one formal charge, now its formal charge is zero, because there are four bonds around it. And this carbon, this carbon over here on the right that had the It's obvious that this carbon here in red has a plus one formal charge, it has three bonds around it. Okay, now it's very obvious, let me point this out in red. So let me draw in those three hydrogens on the right. So we put in a hydrogen here, we put in a hydrogen here, and we put in a hydrogen here. 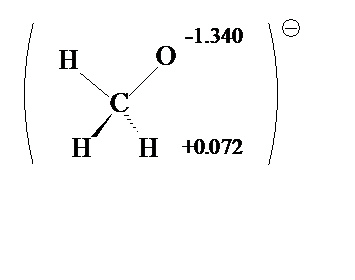
If we draw in our hydrogens, it'll be clear why this is correct. And that moves the positive formal charge over to this carbon. Me make them blue again, the electrons in blue move over to here, like that. You take your electrons, and you move them in the direction of the positive charge, of the positive one formal charge, and so let's show that. So what is the proper resonance structure to draw? Well, let's show that down here. This is not a correct resonance structure. So we added another formal charge, and we have carbon with five bonds, so this is incorrect, If we go to this top carbon here, there's only three bondsĪround that carbon, so that carbon would haveĪ plus one formal charge. There's another reason why this is wrong. In the second period, there's four orbitals, and each orbital can hold a maximum of two electrons, which gives us four times Because of carbon's position on the periodic table, You can't show carbon with five bonds, because that would be 10 electrons around this carbon, and carbon can never exceedĪn octet of electrons. How many bonds are there to that carbon? Well here's one bond, two, three, four, and five, that's five bonds to a carbon, that does not happen, 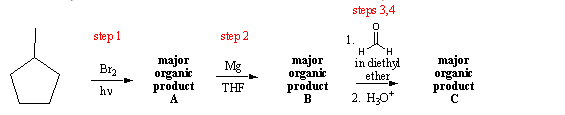
Let's focus in on this carbon right here, the one I marked in red. Why this resonance structure is incorrect. So let's put in those hydrogens for the resonance structure on the right, and it should be obvious Plus one formal charge must have one hydrogen. Has two hydrogens on it, and the carbon with a Has one hydrogen on it, same with this carbon, and this carbon right here Structure not possible? Well, let's draw in the hydrogens on the carbons, and it So the resonance structure on the right, this is an incorrect resonance structure, why is this resonance But this is incorrect, so let me write "no" here. Let me highlight thoseĮlectrons in blue here, so these electrons here move down to here. Some students would take these electrons and move them down to here and say, all right, so on the right, now, I would have this, and this is my resonance structure. So let's say we wanted to draw a resonance structureįor this carbocation. I see a lot of mistakes when students draw resonance structures, and so I wanted to make a video on some of the more common
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
